Why is ether used as the solvent during Grignard reactions?
Responda:
Ether is used as a solvent because it is aprotic and can solvate the magnesium ion.
Explicação:
A Grignard reaction involves the reaction of an alkyl (or aryl halide) with magnesium metal to form an alkylmagnesium halide.
#"R-X" + "Mg" stackrelcolor(blue)("dry ether"color(white)(X))(→) "R-MgX"#
Diethyl ether is an especially good solvent for the formation of Grignard reagents for two reasons.
(a) Ether is an aprotic solvent
O #"R-Mg"# bond is highly polar: #stackrelcolor(blue)(δ^(-))("R"color(white)(l))-stackrelcolor (blue)(δ^+)("Mg")"X"#
The Grignard carbon is highly basic and reacts with the acidic protons of polar solvents like water to form an alkane.
#stackrelcolor (blue)(δ^(-))("R"color(white)(l))"-"stackrelcolor (blue)(δ^+)("Mg")"X" + "H-OH" → "R-H" + "HO-MgX"#
Ether has no acidic protons, so Grignard reagents are stable in ether.
(b) Ether is a great solvating agent
O #"MgX"# bond in a Grignard reagent is ionic: #"R-Mg"^+ color(white)(l)"X"^-#.
Hence, it is difficult to form a Grignard reagent in a nonpolar solvent.
O #"C-O"# bond in an ether is quite polar, and the oxygen of the dipole can solvate and stabilize the #"R-Mg"^+# íon.
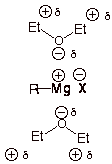
Thus, Grignard reagents are soluble in ether.

